InVivoPlus anti-mouse CD25 (IL-2Rα)
Product Details
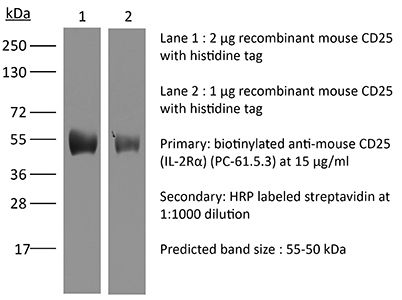
The PC-61.5.3 monoclonal antibody reacts with mouse IL-2Rα also known as CD25, Ly-43, p55, or Tac. IL-2Rα is the 55 kDa ligand-binding subunit of the interleukin 2 receptor alpha chain. IL-2Rα is expressed on activated mature T and B lymphocytes, thymocyte subsets, pre-B cells, and T regulatory cells. IL-2Rα has been shown to play roles in lymphocyte differentiation, activation, and proliferation. Alone, the IL-2Rα binds IL-2 with relatively low affinity however, when IL-2Rα associates with IL-2Rβ (CD122) and the common gamma chain (CD132) the complex binds IL-2 with high affinity. The PC-61.5.3 antibody has been shown to inhibit the binding of IL-2 to both the low and high affinity IL-2 receptor forms. Additionally, the PC-61.5.3 antibody is commonly used to deplete CD4+FoxP3+ T regulatory cells in vivo.Specifications
| Isotype | Rat IgG1, λ |
|---|---|
| Recommended Isotype Control(s) | InVivoPlus rat IgG1 isotype control, anti-horseradish peroxidase |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Immunogen | IL-2-dependent cytolytic mouse T cell clone B6.1 |
| Reported Applications |
in vivo regulatory T cell depletion Flow cytometry |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin* |
<1EU/mg (<0.001EU/μg) Determined by LAL gel clotting assay |
| Sterility | 0.2 μM filtered |
| Production | Purified from tissue culture supernatant in an animal free facility |
| Purification | Protein A High Salt |
| RRID | AB_1107619 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
Additional Formats
Recommended Products
in vivo regulatory T cell depletion
CD4+CD25+Foxp3+ Tregs resolve experimental lung injury in mice and are present in humans with acute lung injury PubMed
Acute lung injury (ALI) is characterized by rapid alveolar injury, inflammation, cytokine induction, and neutrophil accumulation. Although early events in the pathogenesis of ALI have been defined, the mechanisms underlying resolution are unknown. As a model of ALI, we administered intratracheal (i.t.) LPS to mice and observed peak lung injury 4 days after the challenge, with resolution by day 10. Numbers of alveolar lymphocytes increased as injury resolved. To examine the role of lymphocytes in this response, lymphocyte-deficient Rag-1-/- and C57BL/6 WT mice were exposed to i.t. LPS. The extent of injury was similar between the groups of mice through day 4, but recovery was markedly impaired in the Rag-1-/- mice. Adoptive transfer studies revealed that infusion of CD4+CD25+Foxp3+ Tregs as late as 24 hours after i.t. LPS normalized resolution in Rag-1-/- mice. Similarly, Treg depletion in WT mice delayed recovery. Treg transfer into i.t. LPS-exposed Rag-1-/- mice also corrected the elevated levels of alveolar proinflammatory cytokines and increased the diminished levels of alveolar TGF-beta and neutrophil apoptosis. Mechanistically, Treg-mediated resolution of lung injury was abrogated by TGF-beta inhibition. Moreover, BAL of patients with ALI revealed dynamic changes in CD3+CD4+CD25hiCD127loFoxp3+ cells. These results indicate that Tregs modify innate immune responses during resolution of lung injury and suggest potential targets for treating ALI, for which there are no specific therapies currently available.
in vivo regulatory T cell depletion
In situ vaccination combined with androgen ablation and regulatory T-cell depletion reduces castration-resistant tumor burden in prostate-specific pten knockout mice PubMed
There is no effective treatment for prostate cancer arising after androgen ablation. Previous studies have analyzed the short-term effects of androgen ablation on the immune system and suggest an abatement of immune suppression by hormone removal. Because castration-resistant disease can arise years after treatment, it is crucial to determine the duration of immune potentiation by castration. Because immunotherapeutic efficacy is determined by the balance of immune cell subsets and their location within the tumor, we assessed the acute and chronic effect of androgen ablation on the localization of T-cell subsets within castration-resistant murine prostate cancer. We observed a transient increase in CD4+ and CD8+ T-cell numbers at the residual tumor after androgen ablation. More than 2 months later, regulatory T cells (Treg) were increasingly found within prostate epithelium, whereas CTLs, which were evenly distributed before androgen ablation, became sequestered within stroma. Anti-CD25 antibody administration along with castration enhanced CTL access to cancerous glands but did not increase effector function. Intraprostatic injection of LIGHT-expressing tumor cells increased the proportion of CD8+ T cells with functional capacity within the cancerous gland. In addition, Treg depletion within the tumor was enhanced. Together, these manipulations significantly reduced castration-resistant tumor burden. Thus, our results indicate that immune modulations, which prevent Treg accumulation and augment effector cell infiltration of prostatic epithelium, may be effective in reducing tumor burden or preventing tumor recurrence after androgen ablation therapy.
in vivo regulatory T cell depletion
Regulation of induced colonic inflammation by Lactobacillus acidophilus deficient in lipoteichoic acid PubMed
Imbalance in the regulatory immune mechanisms that control intestinal cellular and bacterial homeostasis may lead to induction of the detrimental inflammatory signals characterized in humans as inflammatory bowel disease. Induction of proinflammatory cytokines (i.e., IL-12) induced by dendritic cells (DCs) expressing pattern recognition receptors may skew naive T cells to T helper 1 polarization, which is strongly implicated in mucosal autoimmunity. Recent studies show the ability of probiotic microbes to treat and prevent numerous intestinal disorders, including Clostridium difficile-induced colitis. To study the molecular mechanisms involved in the induction and repression of intestinal inflammation, the phosphoglycerol transferase gene that plays a key role in lipoteichoic acid (LTA) biosynthesis in Lactobacillus acidophilus NCFM (NCK56) was deleted. The data show that the L. acidophilus LTA-negative in LTA (NCK2025) not only down-regulated IL-12 and TNFalpha but also significantly enhanced IL-10 in DCs and controlled the regulation of costimulatory DC functions, resulting in their inability to induce CD4(+) T-cell activation. Moreover, treatment of mice with NCK2025 compared with NCK56 significantly mitigated dextran sulfate sodium and CD4(+)CD45RB(high)T cell-induced colitis and effectively ameliorated dextran sulfate sodium-established colitis through a mechanism that involves IL-10 and CD4(+)FoxP3(+) T regulatory cells to dampen exaggerated mucosal inflammation. Directed alteration of cell surface components of L. acidophilus NCFM establishes a potential strategy for the treatment of inflammatory intestinal disorders.
Flow Cytometry, in vivo CD40 activation, in vivo regulatory T cell depletion
Primary oligodendrocyte death does not elicit anti-CNS immunity PubMed
Anti-myelin immunity is commonly thought to drive multiple sclerosis, yet the initial trigger of this autoreactivity remains elusive. One of the proposed factors for initiating this disease is the primary death of oligodendrocytes. To specifically test such oligodendrocyte death as a trigger for anti-CNS immunity, we inducibly killed oligodendrocytes in an in vivo mouse model. Strong microglia-macrophage activation followed oligodendrocyte death, and myelin components in draining lymph nodes made CNS antigens available to lymphocytes. However, even conditions favoring autoimmunity-bystander activation, removal of regulatory T cells, presence of myelin-reactive T cells and application of demyelinating antibodies-did not result in the development of CNS inflammation after oligodendrocyte death. In addition, this lack of reactivity was not mediated by enhanced myelin-specific tolerance. Thus, in contrast with previously reported impairments of oligodendrocyte physiology, diffuse oligodendrocyte death alone or in conjunction with immune activation does not trigger anti-CNS immunity.
in vivo regulatory T cell depletion
Increased CD8+ T-cell function following castration and immunization is countered by parallel expansion of regulatory T cells PubMed
Although androgen ablation therapy is effective in treating primary prostate cancers, a significant number of patients develop incurable castration-resistant disease. Recent studies have suggested a potential synergy between vaccination and androgen ablation, yet the enhanced T-cell function is transient. Using a defined tumor antigen model, UV-8101-RE, we found that concomitant castration significantly increased the frequency and function of antigen-specific CD8(+) T cells early after the immunization of wild-type mice. However, at a late time point after immunization, effector function was reduced to the same level as noncastrated mice and was accompanied by a concomitant amplification in CD4(+)CD25(+)Foxp3(+) regulatory T cells (Treg) following immunization. We investigated whether Treg expansion occurred following castration of prostate tumor-bearing mice. In the prostate-specific Pten(-/-) mouse model of prostate cancer, we observed an accelerated Treg expansion in mice bearing the castration-resistant endogenous prostate tumor, which prevented effector responses to UV-8101-RE. Treg depletion together with castration elicited a strong CD8(+) T-cell response to UV-8101-RE in Pten(-/-) mice and rescued effector function in castrated and immunized wild-type mice. In addition, Treg expansion in Pten(-/-) mice was prevented by in vivo interleukin (IL)-2 blockade suggesting that increased IL-2 generated by castration and immunization promotes Treg expansion. Our findings therefore suggest that although effector responses are augmented by castration, the concomitant expansion of Tregs is one mechanism responsible for only transient immune potentiation after androgen ablation.
in vivo neutrophil depletion, in vivo NK cell depletion, in vivo regulatory T cell depletion
Macrophage and T cell produced IL-10 promotes viral chronicity PubMed
Chronic viral infections lead to CD8(+) T cell exhaustion, characterized by impaired cytokine secretion. Presence of the immune-regulatory cytokine IL-10 promotes chronicity of Lymphocytic Choriomeningitis Virus (LCMV) Clone 13 infection, while absence of IL-10/IL-10R signaling early during infection results in viral clearance and higher percentages and numbers of antiviral, cytokine producing T cells. IL-10 is produced by several cell types during LCMV infection but it is currently unclear which cellular sources are responsible for induction of viral chronicity. Here, we demonstrate that although dendritic cells produce IL-10 and overall IL-10 mRNA levels decrease significantly in absence of CD11c(+) cells, absence of IL-10 produced by CD11c(+) cells failed to improve the LCMV-specific T cell response and control of LCMV infection. Similarly, NK cell specific IL-10 deficiency had no positive impact on the LCMV-specific T cell response or viral control, even though high percentages of NK cells produced IL-10 at early time points after infection. Interestingly, we found markedly improved T cell responses and clearance of normally chronic LCMV Clone 13 infection when either myeloid cells or T cells lacked IL-10 production and mice depleted of monocytes/macrophages or CD4(+) T cells exhibited reduced overall levels of IL-10 mRNA. These data suggest that the decision whether LCMV infection becomes chronic or can be cleared critically depends on early CD4(+) T cell and monocyte/macrophage produced IL-10.
in vivo blocking of PD-1/PD-L signaling, in vivo regulatory T cell depletion
Impaired selectin-dependent leukocyte recruitment induces T-cell exhaustion and prevents chronic allograft vasculopathy and rejection PubMed
Selectin-selectin ligand interactions mediate the initial steps in leukocyte migration, an integral part of immune responses. Fucosyltransferase-VII (FucT-VII), encoded by Fut7, is essential for biosynthesis of selectin ligands. In an established model of cardiac allograft vasculopathy and chronic rejection, Fut7(-/-) recipients exhibited long-term graft survival with minimal vasculopathy compared with WT controls. Graft survival was associated with CD4 T-cell exhaustion in the periphery, characterized by impaired effector cytokine production, defective proliferation, increased expression of inhibitory receptors programmed death-1 (PD-1) and T cell Ig- and mucin-domain-containing molecule-3 (Tim-3), low levels of IL-7Ralpha on CD4 T cells, and reduced migration of polyfunctional CD4 memory T cells to the allograft. Blocking PD-1 triggered rejection only in Fut7(-/-) recipients, whereas depleting regulatory T cells had no effect in either Fut7(-/-) or WT recipients. Adoptive transfer experiments confirmed that this CD4 T cell-exhausted phenotype is seen primarily in Fut7(-/-) CD4 T cells. These data suggest that impaired leukocyte recruitment is a novel mechanism leading to CD4 T-cell exhaustion. Our experimental system serves as an excellent model to study CD4 T-cell exhaustion as a dominant mechanism of transplant tolerance. Further, targeting FucT-VII may serve as a promising strategy to prevent chronic allograft rejection and promote tolerance.
In vivo CD70 blockade, in vivo regulatory T cell depletion
Dual function of CD70 in viral infection: modulator of early cytokine responses and activator of adaptive responses PubMed
The role of the TNF family member CD70 in adaptive T cell responses has been intensively studied, but its function in innate responses is still under investigation. In this study, we show that CD70 inhibits the early innate response to murine CMV (MCMV) but is essential for the optimal generation of virus-specific CD8 T cells. CD70(-/-) mice reacted to MCMV infection with a robust type I IFN and proinflammatory cytokine response. This response was sufficient for initial control of MCMV, although at later time points, CD70(-/-) mice became more susceptible to MCMV infection. The heightened cytokine response during the early phase of MCMV infection in CD70(-/-) mice was paralleled by a reduction in regulatory T cells (Treg). Treg from naive CD70(-/-) mice were not as efficient at suppressing T cell proliferation compared with Treg from naive wild-type mice, and depletion of Treg during MCMV infection in Foxp3-diphtheria toxin receptor mice or in wild-type mice recapitulated the phenotype observed in CD70(-/-) mice. Our study demonstrates that although CD70 is required for the activation of the antiviral adaptive response, it has a regulatory role in early cytokine responses to viruses such as MCMV, possibly through maintenance of Treg survival and function.
in vivo blocking of CD40/CD40L signaling, in vivo regulatory T cell depletion
Spontaneous restoration of transplantation tolerance after acute rejection PubMed
Transplantation is a cure for end-stage organ failure but, in the absence of pharmacological immunosuppression, allogeneic organs are acutely rejected. Such rejection invariably results in allosensitization and accelerated rejection of secondary donor-matched grafts. Transplantation tolerance can be induced in animals and a subset of humans, and enables long-term acceptance of allografts without maintenance immunosuppression. However, graft rejection can occur long after a state of transplantation tolerance has been acquired. When such an allograft is rejected, it has been assumed that the same rules of allosensitization apply as to non-tolerant hosts and that immunological tolerance is permanently lost. Using a mouse model of cardiac transplantation, we show that when Listeria monocytogenes infection precipitates acute rejection, thus abrogating transplantation tolerance, the donor-specific tolerant state re-emerges, allowing spontaneous acceptance of a donor-matched second transplant. These data demonstrate a setting in which the memory of allograft tolerance dominates over the memory of transplant rejection.
Flow Cytometry, in vivo regulatory T cell depletion
PD-1 upregulated on regulatory T cells during chronic virus infection enhances the suppression of CD8+ T cell immune response via the interaction with PD-L1 expressed on CD8+ T cells PubMed
Regulatory T (Treg) cells act as terminators of T cell immuniy during acute phase of viral infection; however, their role and suppressive mechanism in chronic viral infection are not completely understood. In this study, we compared the phenotype and function of Treg cells during acute or chronic infection with lymphocytic choriomeningitis virus. Chronic infection, unlike acute infection, led to a large expansion of Treg cells and their upregulation of programmed death-1 (PD-1). Treg cells from chronically infected mice (chronic Treg cells) displayed greater suppressive capacity for inhibiting both CD8(+) and CD4(+) T cell proliferation and subsequent cytokine production than those from naive or acutely infected mice. A contact between Treg and CD8(+) T cells was necessary for the potent suppression of CD8(+) T cell immune response. More importantly, the suppression required cell-specific expression and interaction of PD-1 on chronic Treg cells and PD-1 ligand on CD8(+) T cells. Our study defines PD-1 upregulated on Treg cells and its interaction with PD-1 ligand on effector T cells as one cause for the potent T cell suppression and proposes the role of PD-1 on Treg cells, in addition to that on exhausted T cells, during chronic viral infection.
Flow Cytometry, in vivo LFA-1 neutralization, in vivo regulatory T cell depletion
Integrin alpha L controls the homing of regulatory T cells during CNS autoimmunity in the absence of integrin alpha 4 PubMed
Experimental autoimmune encephalomyelitis (EAE), the animal model of multiple sclerosis (MS), results from an autoimmune attack of the central nervous system (CNS) by effector T helper (Th) 1 and Th17 cells. Regulatory T cells (Treg) can control effector T cells and limit the progression of CNS autoimmunity. Integrin alpha 4 (Itga4) is critical for the entry of Th1 but not Th17 cells into the CNS during EAE. Whether Itga4 controls the homing of Tregs in the CNS and whether Tregs can limit Th17-mediated EAE has, however, not been addressed. Through selective elimination of Itga4 in Foxp3-expressing cells, we show here that Tregs can suppress Th17-mediated EAE and enter into the CNS independently of Itga4. Furthermore, similarly to Th17 cells and in contrast to Th1 cells, Tregs depend on LFA-1 for their entry into the CNS in the absence of Itga4. Therefore, these data suggest that the efficacy of Itga4 neutralization on MS progression may be associated with the prevention of Th1 cells and the maintenance of Tregs migration into the CNS.
in vivo blocking of IL-10/IL-10R signaling, in vivo CD4+ T cell depletion, in vivo CD8+ T cell depletion, in vivo regulatory T cell depletion, in vivo TNFα neutralization
Depletion of regulatory T cells in a hapten-induced inflammation model results in prolonged and increased inflammation driven by T cells PubMed
Regulatory T cells (Tregs ) are known to play an immunosuppressive role in the response of contact hypersensitivity (CHS), but neither the dynamics of Tregs during the CHS response nor the exaggerated inflammatory response after depletion of Tregs has been characterized in detail. In this study we show that the number of Tregs in the challenged tissue peak at the same time as the ear-swelling reaches its maximum on day 1 after challenge, whereas the number of Tregs in the draining lymph nodes peaks at day 2. As expected, depletion of Tregs by injection of a monoclonal antibody to CD25 prior to sensitization led to a prolonged and sustained inflammatory response which was dependent upon CD8 T cells, and co-stimulatory blockade with cytotoxic T lymphocyte antigen-4-immunoglobulin (CTLA-4-Ig) suppressed the exaggerated inflammation. In contrast, blockade of the interleukin (IL)-10-receptor (IL-10R) did not further increase the exaggerated inflammatory response in the Treg -depleted mice. In the absence of Tregs , the response changed from a mainly acute reaction with heavy infiltration of neutrophils to a sustained response with more chronic characteristics (fewer neutrophils and dominated by macrophages). Furthermore, depletion of Tregs enhanced the release of cytokines and chemokines locally in the inflamed ear and augmented serum levels of the systemic inflammatory mediators serum amyloid (SAP) and haptoglobin early in the response.
Flow Cytometry, in vivo IFNγ neutralization, in vivo IL-12 neutralization, in vivo regulatory T cell depletion
Anti-CD20 therapy induces a memory Th1 response through the IFN-gamma/IL-12 axis and prevents protumor regulatory T-cell expansion in mice PubMed
The long-lasting clinical response by lymphoma patients to anti-CD20 therapy has been attributed to the induction of an anti-tumor adaptive immunity. We previously demonstrated that a CD4-dependent mechanism is responsible for the long-term protection of CD20(+) tumor-bearing mice by anti-CD20 treatment. Here, we compare tumor immunity in tumor-bearing animals that did or did not receive anti-CD20 treatment. Splenic CD4(+)FoxP3(+) regulatory T cells (Tregs) expanded substantially in untreated mice that exhibited then a reduced survival, whereas Tregs depletion led to long-term survival of the animals, suggesting the establishment of a Treg-dependent immunosuppressive environment after tumor injection. Strikingly, anti-CD20 therapy reversed the initial expansion of Tregs, and was accompanied by a marked increase in the number of Th1 cells, with no detectable change in Th2 and Th17 cell numbers. Interleukin-12 serum level was also increased by the anti-CD20 treatment, and activated myeloid dendritic cells producing interleukin-12 could be detected in lymph nodes of treated animals, while interferon-gamma blockade strongly reduced survival. Also, CD4(+) effector memory T cells were evidenced in surviving animals, and the transfer of CD4(+) T cells induced long-term protection. Thus, anti-CD20 therapy promotes strong anti-tumor adaptive immunity, opposes Treg expansion and inhibits tumor cells from maintaining an immunosuppressive environment.
in vivo IFNγ neutralization, in vivo IL-10 neutralization, in vivo IL-21R blockade, in vivo regulatory T cell depletion, in vivo TGFβ neutralization
Expanding antigen-specific regulatory networks to treat autoimmunity PubMed
Regulatory T cells hold promise as targets for therapeutic intervention in autoimmunity, but approaches capable of expanding antigen-specific regulatory T cells in vivo are currently not available. Here we show that systemic delivery of nanoparticles coated with autoimmune-disease-relevant peptides bound to major histocompatibility complex class II (pMHCII) molecules triggers the generation and expansion of antigen-specific regulatory CD4(+) T cell type 1 (TR1)-like cells in different mouse models, including mice humanized with lymphocytes from patients, leading to resolution of established autoimmune phenomena. Ten pMHCII-based nanomedicines show similar biological effects, regardless of genetic background, prevalence of the cognate T-cell population or MHC restriction. These nanomedicines promote the differentiation of disease-primed autoreactive T cells into TR1-like cells, which in turn suppress autoantigen-loaded antigen-presenting cells and drive the differentiation of cognate B cells into disease-suppressing regulatory B cells, without compromising systemic immunity. pMHCII-based nanomedicines thus represent a new class of drugs, potentially useful for treating a broad spectrum of autoimmune conditions in a disease-specific manner.
in vivo regulatory T cell depletion
A T cell-specific deletion of HDAC1 protects against experimental autoimmune encephalomyelitis PubMed
Multiple sclerosis (MS) is a human neurodegenerative disease characterized by the invasion of autoreactive T cells from the periphery into the CNS. Application of pan-histone deacetylase inhibitors (HDACi) ameliorates experimental autoimmune encephalomyelitis (EAE), an animal model for MS, suggesting that HDACi might be a potential therapeutic strategy for MS. However, the function of individual HDAC members in the pathogenesis of EAE is not known. In this study we report that mice with a T cell-specific deletion of HDAC1 (using the Cd4-Cre deleter strain; HDAC1-cKO) were completely resistant to EAE despite the ability of HDAC1cKO CD4(+) T cells to differentiate into Th17 cells. RNA sequencing revealed STAT1 as a prominent upstream regulator of differentially expressed genes in activated HDAC1-cKO CD4(+) T cells and this was accompanied by a strong increase in phosphorylated STAT1 (pSTAT1). This suggests that HDAC1 controls STAT1 activity in activated CD4(+) T cells. Increased pSTAT1 levels correlated with a reduced expression of the chemokine receptors Ccr4 and Ccr6, which are important for the migration of T cells into the CNS. Finally, EAE susceptibility was restored in WT:HDAC1-cKO mixed BM chimeric mice, indicating a cell-autonomous defect. Our data demonstrate a novel pathophysiological role for HDAC1 in EAE and provide evidence that selective inhibition of HDAC1 might be a promising strategy for the treatment of MS.