Fc-Free or Full IgG? How Antibody Format Shapes In Vivo Data
Antibody format is not a passive variable. In mechanistic in vivo studies, the choice between a full IgG, a Fab, and an F(ab')₂ determines which biology you are measuring and which biology may be unintentionally introduced.
The Result You Did Not Design For
You have designed a clean study. The target is well defined, the model is validated, and the readout is appropriately sensitive. And yet the data look off. The response is stronger than expected, or absent where theory predicts presence. The pattern resists straightforward interpretation.
This is a familiar scenario in mechanistic in vivo work. The instinct is to interrogate the target biology: receptor expression, model fidelity, dosing, and timing. Rarely does the question turn to the antibody itself, not to the variable domain, but to the portion of the molecule often assumed to be functionally neutral.
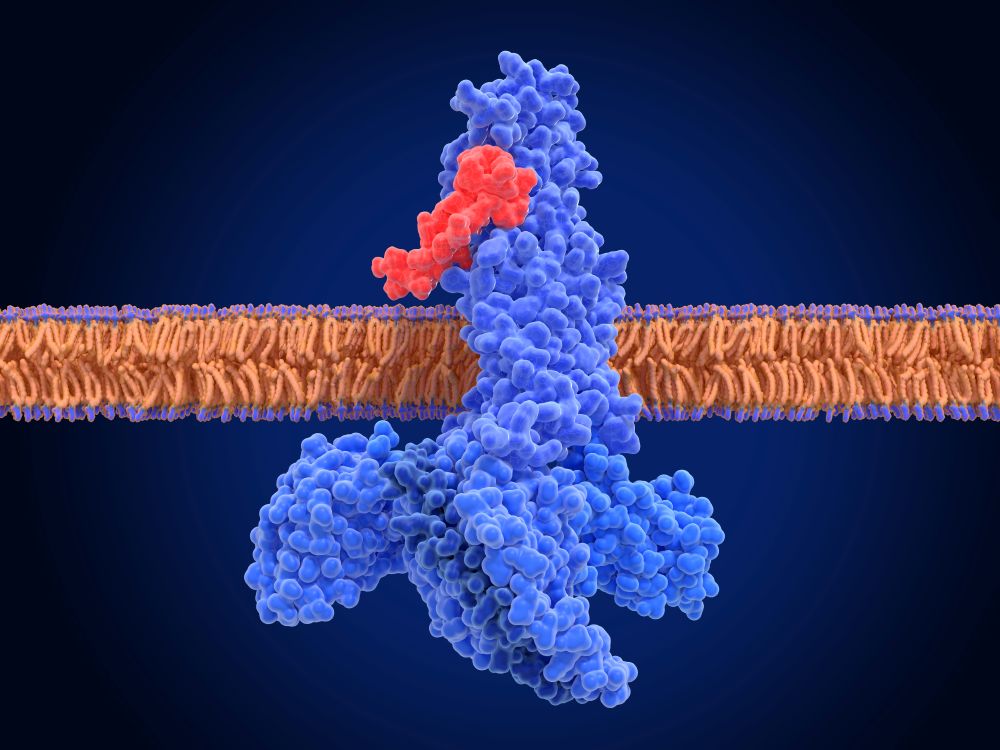
The Fc region is often treated as an inert scaffold. In practice, it is not.
The Fc Region as an Active Biological Component
The fragment crystallizable (Fc) domain of an IgG molecule provides structural stability and contributes to serum half-life through neonatal Fc receptor (FcRn) recycling. It also engages Fc gamma receptors (FcγRs) expressed across immune cell populations and can activate complement pathways through interaction with C1q in certain contexts.
These interactions are not incidental. FcγRs are expressed on macrophages, monocytes, and NK cells, among other immune cell types. Their engagement can initiate downstream signaling pathways that drive antibody-dependent cellular phagocytosis (ADCP) and antibody-dependent cellular cytotoxicity (ADCC). These effector functions are intentionally leveraged in therapeutic antibody design but may not be part of the biological question being studied in mechanistic experiments.1, 2
Importantly, FcγR signaling is context-dependent. Receptor subtype, expression level, and tissue distribution can influence how Fc engagement contributes to the observed response.
What Fc Engagement Can Produce In Vivo
Fc-mediated engagement can activate multiple immune pathways depending on the model and context.
Macrophages expressing Fcγ receptors can mediate phagocytosis of antibody-bound targets, contributing to target cell clearance. Weiskopf and Weissman demonstrated that macrophage-mediated ADCP has been shown to be a primary mechanism of action for multiple approved cancer antibodies, illustrating that this pathway can operate at scale in vivo.3
Natural killer cells, through FcγRIIIa (CD16), can induce cytotoxic responses against antibody-coated cells. Campbell et al. documented FcγRIIIa-mediated NK cell activation as a central component of elotuzumab’s mechanism in multiple myeloma, illustrating how CD16 engagement can drive robust ADCC in vivo.4
Complement pathways may also be engaged through Fc-C1q interactions, contributing to downstream inflammatory signaling and, in certain contexts, direct cell lysis.5
Together, these mechanisms mean that in an in vivo study using a full IgG, the observed response may reflect both target-driven biology and Fc-mediated immune activity.
When Format Becomes a Source of Interpretive Complexity
The practical consequence of Fc engagement is that it can introduce additional biological signals beyond the intended mechanism under study.
In checkpoint or agonist antibody studies, for example, observed activity may be influenced by FcγR-mediated crosslinking in addition to direct receptor engagement. In TNFR superfamily systems, receptor clustering is often required for signaling, and antibody structure can influence whether that clustering occurs.
Richards et al. demonstrated that engineered ligand-mimetic constructs can drive receptor activation independent of FcγR engagement, highlighting how format and structure can influence signaling outcomes in systems where clustering is required.6
These effects are not inherently problematic. However, when Fc-mediated contributions are not explicitly considered, they can complicate interpretation of whether the observed response is driven by the intended pathway.
Antibody Format as an Experimental Control Variable
Reframing antibody format as an experimental design choice, rather than a default, changes how these variables are managed.
- Full IgG: Bivalent, Fc present. Enables FcγR engagement, complement activation, and extended half-life. Appropriate when effector function or cell depletion is part of the experimental objective.
- Fab (~50 kDa): Monovalent, Fc absent. Eliminates Fc-mediated interactions and prevents receptor crosslinking. Appropriate for blocking studies where monovalent engagement is required.
- F(ab')₂ (~110 kDa): Bivalent, Fc absent. Preserves avidity and receptor crosslinking while removing Fc-driven effector functions. Appropriate when clustering-dependent signaling is required without FcγR contribution.
The distinction between Fab and F(ab')₂ is particularly important in systems where receptor clustering influences signaling. For certain receptor families, including members of the TNFR superfamily, multimerization is often required for productive signaling.
A Practical Framework for Format Selection
Use a full IgG when:
- Effector function, including ADCC or ADCP, is part of the mechanism being studied
- Target cell depletion is an intended outcome
- Fc biology is explicitly included and controlled
Use a Fab when:
- Blocking without receptor crosslinking is required
- Fc-mediated effects need to be removed
- Monovalent engagement is necessary
Use an F(ab')₂ when:
- Receptor clustering is required
- Fc-mediated effects need to be removed
- A format-matched comparison to IgG is needed
Conclusion
Antibody format determines more than binding. It defines which biological mechanisms may be active within an experimental system.
When Fc-mediated interactions are not considered as part of study design, they may contribute to observed outcomes in ways that are not immediately apparent. Treating format as a controllable variable allows for more precise alignment between experimental design and biological interpretation.
Beyond mechanistic studies, antibody format can also influence how molecules behave in more complex systems, including delivery and cell-targeting workflows.
References
1. Liu R, Oldham RJ, Teal E, Beers SA, Cragg MS. Fc-Engineering for Modulated Effector Functions: Improving Antibodies for Cancer Treatment. Antibodies (Basel). 2020;9(4):64. Open access article
2. Vidarsson G, Dekkers G, Rispens T. IgG subclasses and allotypes: from structure to effector functions. Frontiers in Immunology. 2014;5:520. Open access article
3. Weiskopf K, Weissman IL. Macrophages are critical effectors of antibody therapies for cancer. mAbs. 2015;7(2):303-310. Open access article
4. Campbell KS, Cohen AD, Pazina T. Mechanisms of NK Cell Activation and Clinical Activity of the Therapeutic SLAMF7 Antibody, Elotuzumab in Multiple Myeloma. Frontiers in Immunology. 2018;9:2551. Open access article
5. van Erp EA, Luytjes W, Ferwerda G, van Kasteren PB. Fc-Mediated Antibody Effector Functions During Respiratory Syncytial Virus Infection and Disease. Frontiers in Immunology. 2019;10:548. Open access article
6. Richards DM, Marschall V, Billian-Frey K, et al. HERA-GITRL activates T cells and promotes anti-tumor efficacy independent of FcγR-binding functionality. Journal for ImmunoTherapy of Cancer. 2019;7(1):191. Open access article