InVivoMAb anti-mouse CD3
Product Description
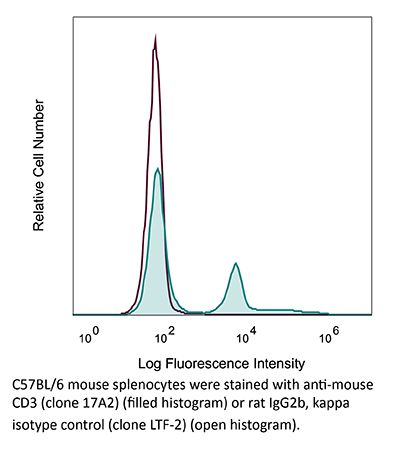
Specifications
| Isotype | Rat IgG2b, κ |
|---|---|
| Recommended Isotype Control(s) | InVivoMAb rat IgG2b isotype control, anti-keyhole limpet hemocyanin |
| Recommended Dilution Buffer | InVivoPure pH 7.0 Dilution Buffer |
| Conjugation | This product is unconjugated. Conjugation is available via our Antibody Conjugation Services. |
| Immunogen | γδ TCR-positive T-T hybridoma D1 |
| Reported Applications | in vitro T cell stimulation/activation |
| Formulation |
PBS, pH 7.0 Contains no stabilizers or preservatives |
| Endotoxin |
≤1EU/mg (≤0.001EU/μg) Determined by LAL assay |
| Purity |
≥95% Determined by SDS-PAGE |
| Sterility | 0.2 µm filtration |
| Production | Purified from cell culture supernatant in an animal-free facility |
| Purification | Protein G |
| RRID | AB_1107630 |
| Molecular Weight | 150 kDa |
| Storage | The antibody solution should be stored at the stock concentration at 4°C. Do not freeze. |
| Need a Custom Formulation? | See All Antibody Customization Options |
Application References
-
Shen, P. X., et al (2021). "Urolithin A ameliorates experimental autoimmune encephalomyelitis by targeting aryl hydrocarbon receptor" EBioMedicine 64: 103227.
PubMed
BACKGROUND: Urolithin A (URA) is an intestinal microbiota metabolic product from ellagitannin-containing foods with multiple biological activities. However, its role in autoimmune diseases is largely unknown. Here, for first time, we demonstrate the therapeutic effect of URA in an experimental autoimmune encephalomyelitis (EAE) animal model. METHODS: Therapeutic effect was evaluated via an active and passive EAE animal model in vivo. The function of URA on bone marrow-derived dendritic cells (BM-DCs), T cells, and microglia were tested in vitro. FINDINGS: Oral URA (25 mg/kg/d) suppressed disease progression at prevention, induction, and effector phases of preclinical EAE. Histological evaluation showed that significantly fewer inflammatory cells, decreased demyelination, lower numbers of M1-type microglia and activated DCs, as well as reduced infiltrating Th1/Th17 cells were present in the central nervous system (CNS) of the URA-treated group. URA treatment at 25 μM inhibited the activation of BM-DCs in vitro, restrained Th17 cell differentiation in T cell polarization conditions, and in a DC-CD4(+) T cell co-culture system. Moreover, we confirmed URA inhibited pathogenicity of Th17 cells in adoptive EAE. Mechanism of URA action was directly targeting Aryl Hydrocarbon Receptor (AhR) and modulating the signaling pathways. INTERPRETATION: Collectively, our study offers new evidence that URA, as a human microbial metabolite, is valuable to use as a prospective therapeutic candidate for autoimmune diseases.
-
Edwards-Hicks, J., et al (2020). "Metabolic Dynamics of In Vitro CD8+ T Cell Activation" Metabolites 11(1).
PubMed
CD8+ T cells detect and kill infected or cancerous cells. When activated from their naïve state, T cells undergo a complex transition, including major metabolic reprogramming. Detailed resolution of metabolic dynamics is needed to advance the field of immunometabolism. Here, we outline methodologies that when utilized in parallel achieve broad coverage of the metabolome. Specifically, we used a combination of 2 flow injection analysis (FIA) and 3 liquid chromatography (LC) methods in combination with positive and negative mode high-resolution mass spectrometry (MS) to study the transition from naïve to effector T cells with fine-grained time resolution. Depending on the method, between 54% and 98% of measured metabolic features change in a time-dependent manner, with the major changes in both polar metabolites and lipids occurring in the first 48 h. The statistical analysis highlighted the remodeling of the polyamine biosynthesis pathway, with marked differences in the dynamics of precursors, intermediates, and cofactors. Moreover, phosphatidylcholines, the major class of membrane lipids, underwent a drastic shift in acyl chain composition with polyunsaturated species decreasing from 60% to 25% of the total pool and specifically depleting species containing a 20:4 fatty acid. We hope that this data set with a total of over 11,000 features recorded with multiple MS methodologies for 9 time points will be a useful resource for future work.
-
Nance, J. P., et al (2015). "Bcl6 middle domain repressor function is required for T follicular helper cell differentiation and utilizes the corepressor MTA3" Proc Natl Acad Sci U S A. pii : 201507312.
PubMed
T follicular helper (Tfh) cells are essential providers of help to B cells. The transcription factor B-cell CLL/lymphoma 6 (Bcl6) is a lineage-defining regulator of Tfh cells and germinal center B cells. In B cells, Bcl6 has the potential to recruit distinct transcriptional corepressors through its BTB domain or its poorly characterized middle domain (also known as RDII), but in Tfh cells the roles of the Bcl6 middle domain have yet to be clarified. Mimicked acetylation of the Bcl6 middle domain (K379Q) in CD4 T cells results in significant reductions in Tfh differentiation in vivo. Blimp1 (Prdm1) is a potent inhibitor of Tfh cell differentiation. Although Bcl6 K379Q still bound to the Prdm1 cis-regulatory elements in Tfh cells, Prdm1 expression was derepressed. This was a result of the failure of Bcl6 K379Q to recruit metastasis-associated protein 3 (MTA3). The loss of Bcl6 function in Bcl6 K379Q-expressing CD4 T cells could be partially rescued by abrogating Prdm1 expression. In addition to Prdm1, we found that Bcl6 recruits MTA3 to multiple genes involved in Tfh cell biology, including genes important for cell migration, cell survival, and alternative differentiation pathways. Thus, Bcl6 middle domain mediated repression is a major mechanism of action by which Bcl6 controls CD4 T-cell fate and function.
-
Hu, S., et al (2014). "Activated CD8+ T lymphocytes inhibit neural stem/progenitor cell proliferation: role of interferon-gamma" PLoS One 9(8): e105219.
PubMed
The ability of neural stem/progenitor cells (NSCs) to self-renew, migrate to damaged sites, and differentiate into neurons has renewed interest in using them in therapies for neurodegenerative disorders. Neurological diseases, including viral infections of the brain, are often accompanied by chronic inflammation, whose impact on NSC function remains unexplored. We have previously shown that chronic neuroinflammation, a hallmark of experimental herpes simplex encephalitis (HSE) in mice, is dominated by brain-infiltrating activated CD8 T-cells. In the present study, activated CD8 lymphocytes were found to suppress NSC proliferation profoundly. Luciferase positive (luc+) NSCs co-cultured with activated, MHC-matched, CD8+ lymphocytes (luc-) showed two- to five-fold lower luminescence than co-cultures with un-stimulated lymphocytes. On the other hand, similarly activated CD4+ lymphocytes did not suppress NSC growth. This differential lymphocyte effect on proliferation was confirmed by decreased BrdU uptake by NSC cultured with activated CD8 T-cells. Interestingly, neutralizing antibodies to interferon-gamma (IFN-gamma) reversed the impact of CD8 lymphocytes on NSCs. Antibodies specific to the IFN-gamma receptor-1 subunit complex abrogated the inhibitory effects of both CD8 lymphocytes and IFN-gamma, indicating that the inhibitory effect of these cells was mediated by IFN-gamma in a receptor-specific manner. In addition, activated CD8 lymphocytes decreased levels of nestin and Sox2 expression in NSCs while increasing GFAP expression, suggesting possible induction of an altered differentiation state. Furthermore, NSCs obtained from IFN-gamma receptor-1 knock-out embryos were refractory to the inhibitory effects of activated CD8+ T lymphocytes on cell proliferation and Sox2 expression. Taken together, the studies presented here demonstrate a role for activated CD8 T-cells in regulating NSC function mediated through the production of IFN-gamma. This cytokine may influence neuro-restorative processes and ultimately contribute to the long-term sequelae commonly seen following herpes encephalitis.
Product Citations
-
A fungi-derived cyclic peptide enhances Th9-mediated antitumor immunity by targeting ZAP70 and SREBP1.
In J Clin Invest on 2 February 2026 by Zhao, W., Zhou, Y., et al.
PubMed
Adoptive cell therapy (ACT) relies on durable and functional T cells to mediate tumor clearance. Th9 cells are a metabolically fit CD4+ T cell subset with strong persistence but limited cytotoxicity. Here, we identified endomelipeptide A (EpA), a cyclic peptide isolated from Ganoderma lucidum-associated endophytic fungi, as a potent enhancer of Th9 cell differentiation. EpA promoted a cytotoxic Th9 phenotype with enhanced mitochondrial function and metabolic fitness. Mechanistically, EpA dually targeted ZAP70 and SREBP1, coupling T cell receptor signaling activation with lipid metabolism suppression. EpA-treated Th9 cells mediated robust, CD8+ T cell-dependent tumor control and enhanced the efficacy of human Th9 CAR T cell therapy in vivo. These findings establish EpA as a distinct cyclic peptide that reprograms Th9 cells and provides a potential approach to boost ACT efficacy.
-
Inflammation in a severe model of dystrophic cardiomyopathy contains a high proportion of T cells that contribute to onset of pathology.
In Am J Physiol Heart Circ Physiol on 1 February 2026 by Piepho, A. B., Krishna, S., et al.
PubMed
Duchenne muscular dystrophy (DMD) is a severe neuromuscular disorder with progressive muscle degeneration and cardiomyopathy leading to heart failure. The inflammatory environment in dystrophic skeletal muscle is well-studied, but little is known about inflammation in DMD cardiomyopathy due to the lack of adequate animal models. We recently developed the Fiona/dko mouse model, deficient for both dystrophin and utrophin, but containing a skeletal muscle-specific expressing utrophin transgene allowing progression of dystrophic cardiomyopathy. This Fiona/dko model is the first DMD cardiomyopathy model to reproducibly progress to reduced cardiac contractile function by 9 mo. In this study, we compared immune cell composition between Fiona/dko mice and their milder littermates that develop cardiac pathology, but do not demonstrate whole heart dysfunction. Flow cytometry analysis revealed that T cells constitute a significant proportion of the immune cell population in dystrophic hearts, in contrast to the known predominantly myeloid signature in dystrophic skeletal muscles. T cell infiltration precedes the development of cardiac fibrosis and dysfunction in Fiona/dko mice. RNA sequencing of whole hearts after cardiac dysfunction shows increased expression of 68 genes related to T cell signaling in Fiona/dko compared with their milder littermates. Furthermore, depletion of circulating CD3+ T cells with a neutralizing antibody ameliorates early pathology in Fiona/dko hearts. Together, these data suggest a role for T cells in the initiation and persistence of dystrophic cardiomyopathy. These findings highlight the distinct inflammatory environment in the dystrophic heart and provide new insights into DMD cardiomyopathy, paving the way for the future development of targeted anti-inflammatory therapies.NEW & NOTEWORTHY Heart failure has become the leading cause of death in Duchenne muscular dystrophy, a progressive degenerative disease of striated muscles. This study highlights an inflammatory environment in dystrophic heart with a high proportion of T-cells that is distinct from the predominant myeloid inflammation in dystrophic skeletal muscles. T-cell-related signaling is associated with the severity of cardiomyopathy, and T-cells contribute to dystrophic cardiomyopathy onset. This data will inform optimal patient treatments that target cardiac inflammation.
-
An engineered human hepatitis A virus capable of rapid proliferation in vitro and causing hepatitis in mice.
In JHEP Rep on 1 December 2025 by Li, J., Jiang, P. Y., et al.
PubMed
Hepatitis A virus (HAV) remains a significant public health threat. The HM175-mp4-based mouse model has advanced pathogenesis research, but its slow in vitro replication limits reverse genetic studies. We aimed to develop a genetically tractable HAV model through rational mutagenesis.
-
Harnessing the dual immunomodulatory function of myeloid-derived suppressor cells to reshape the inflammatory microenvironment for osteoarthritis therapy.
In Mater Today Bio on 1 December 2025 by Guo, Z., Chen, T., et al.
PubMed
Osteoarthritis (OA) pathogenesis is profoundly influenced by dysregulated immune dynamics, where persistent interleukin-17 (IL-17)/T helper 17 (Th17) cell mediated inflammation coordinates with failed regenerative processes to perpetuate joint destruction. Here, we unveil the role of myeloid-derived suppressor cells (MDSCs) as dual-phase regulators that paradoxically orchestrate both inflammatory escalation and tissue repair in OA progression. Intra-articular administration of MDSCs in OA mice amplified IL-17 dependent inflammatory cascades and chemokine-driven leukocyte recruitment, revealing a context-dependent pro-inflammatory phenotype. Unexpectedly, MDSC depletion failed to attenuate joint damage, implying their indispensable yet multifaceted role in OA pathogenesis. Mechanistically, MDSCs exhibited functional plasticity by upregulating arginase-1 to polarize M2 macrophages, fostering a regenerative niche alongside their inflammatory activity. To resolve this duality, we developed a bio-responsive hydrogel-microsphere system integrating transforming growth factor β1 (TGF-β1) and interleukin-1 β1 antibody (anti-IL-1β) loaded mesoporous silica nanoparticles (MSNs). This spatiotemporally controlled platform selectively suppressed MDSC-mediated Th17 cell expansion while harnessing their intrinsic capacity to drive M2 macrophage polarization and chondrogenesis. The resultant shift from a pro-inflammatory to pro-regenerative microenvironment significantly attenuated cartilage erosion and restored joint integrity in OA models. Our findings redefine MDSCs as bifunctional immune orchestrators in OA and establish precision biomaterial guided immune decoding as a paradigm-shifting therapeutic strategy. By engineering MDSCs plasticity through antagonistic cytokine delivery, this work provides a blueprint for microenvironment remodeling in degenerative joint diseases.